The hole/electron carriers introduced by elemental substitutions, intercalations of anion/cation species between the two dimensional (2D) layers, or the gate-bias in field-effect-transistor (FET) structures can make carrier doped situations and induce anomalous electronic states 12, 13. In the Mott insulating systems, relatively strong magnetic interactions with J/k B = −10 2–4 K originating from the on-site and the inter-site Coulomb correlations are playing substantial roles 10, 11. Tasaki proposed another type of ferromagnetism in Hubbard system by taking a next-nearest-hopping into account 9. Similar result was reported by Penn almost the same time 8. Nagaoka proposed ferromagnetism in a quite lightly doped Mott insulator in 1966, where only one hole is added to a Mott-insulator 7. Although microscopic mechanisms for ferromagnetic interaction are limited to several types so far 1, 2, 3, 4, 5, 6, theoretical proposals for different types of ferromagnets have been made analytically in Hubbard Hamiltonian. The role of ferromagnets becomes more and more important as the electronics/spintronics and motorization technologies are advanced. The realization of ferromagnetic state in Nagaoka–Penn mechanism can paves a way for designing new molecules-based ferromagnets in future. We also report emergence of metallic states through further doping and applying external pressures for this doping induced ferromagnetic state. The variation of the magnitude of net polarizations by using the difference of counter cations revealed the systematic change of the ground state from antiferromagnetic one to ferromagnetic one. Heat capacity, magnetization, and ESR measurements substantiated the formation of a bulk ferromagnetic state around 1.0 K with quite soft magnetization versus magnetic field ( M– H) characteristics in (Et-4BrT) 2 where Et-4BrT is ethyl-4-bromothiazolium. A series of Ni(dmit) 2 anion radical salts with organic cations, where dmit is 1,3-dithiole-2-thione-4,5-dithiolate can form bi-layer structure with polarized cation layers. 
We report the first experimental possibility of such ferromagnetism in a molecular Mott insulator with an extremely light and homogeneous hole-doping in π-electron layers induced by net polarization of counterions. Although theoretical possibility of a ferromagnetic ground state has been suggested by Nagaoka and Penn as single-hole doping in a Mott insulator, experimental realization has not been reported more than half century. Magnetic properties of Mott–Hubbard systems are generally dominated by strong antiferromagnetic interactions produced by the Coulomb repulsion of electrons. Textbook content produced by OpenStax is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. 
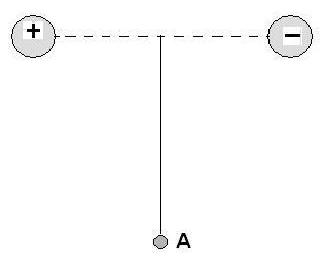
We recommend using aĪuthors: John McMurry, Professor Emeritus Use the information below to generate a citation. Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses theĬreative Commons Attribution-NonCommercial-ShareAlike License Thus, if one positive charge and one negative charge are separated by 100 pm (a bit less than the length of a typical covalent bond), the dipole moment is 1.60 × 10 –29 C For example, the unit charge on an electron is 1.60 × 10 –19 C. Dipole moments are expressed in debyes (D), where 1 D = 3.336 × 10 –30 coulomb meters (C The dipole moment, μ (lowercase Greek letter mu), is defined as the magnitude of the charge Q at either end of the molecular dipole times the distance r between the charges, μ = Q × r. If these two centers don’t coincide, then the molecule has a net polarity. Net polarity is measured by a quantity called the dipole moment and can be thought of in the following way: assume that there is a center of mass of all positive charges (nuclei) in a molecule and a center of mass of all negative charges (electrons). As a practical matter, strongly polar substances are often soluble in polar solvents like water, whereas less polar substances are insoluble in water. Molecular polarity results from the vector summation of all individual bond polarities and lone-pair contributions in the molecule. Just as individual bonds are often polar, molecules as a whole are often polar as well. Polar Covalent Bonds and Dipole Moments. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
